CombiMax
For the symptomatic treatment of osteoarthritis in the knee
CombiMax
-
Na-hyaluronate 36mg/2.25ml
-
Chondroitin sulfate 67.5mg/2.25ml
-
N-acetylglucosamine (NAG) 67.5mg/2.25ml
Sterile, viscoelastic solution for intra-articular use indicated for the symptomatic treatment of osteoarthritis in the knee.

Composition:
It consists of crossed chains of sodium hyaluronate, of a medium molecular weight (3 x 106 Dalton) obtained by fermentation of the bacterial strain Streptococcus and chondroitin sodium sulfate obtained from beef cartilage and NAG obtained from chitin, a natural polymer of long chain N-acetylglucosamine.
This molecular weight promotes strong binding to fibroblast receptors, activation of a large number of receptors and stimulation of endogenous biosynthesis of hyaluronic acid.
The crosslinking of the chains improves the viscoelastic properties of sodium hyaluronate and provides greater resistance to enzymatic, oxidative, thermal and mechanical damage.
Method of administration:
The product is administered exclusively as an intra-articular injection.
Do not inject the product extra-articularly.
A positive therapeutic effect after one administered injection can last up to a year.
After the injection, resting is recommended for 24 hours, in order to further influence the long-term therapeutic effect.
After a period of 6 months, the therapy can be repeated.
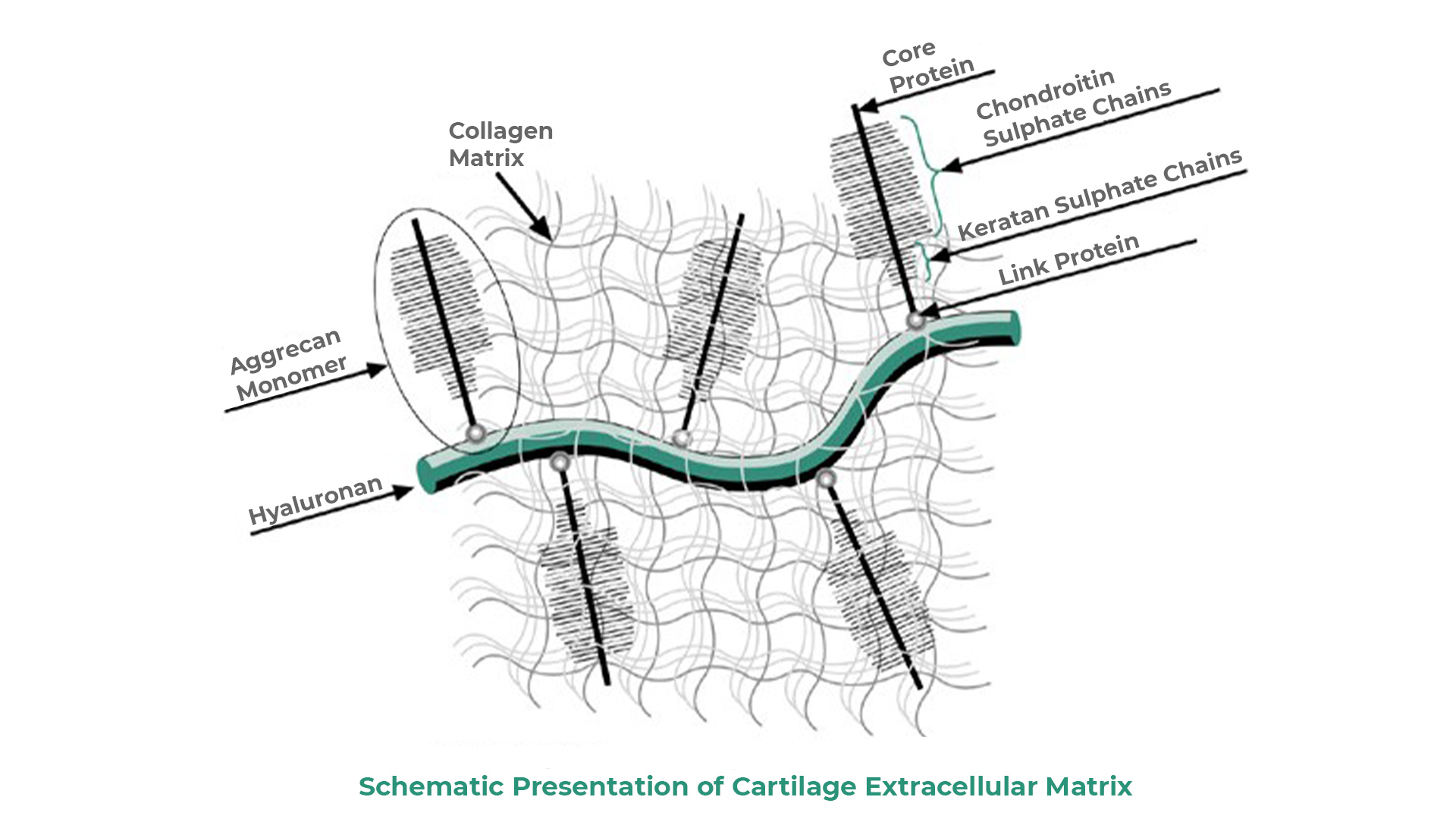
Hyaluronic acid
Hyaluronic acid is a typical glycosaminoglycan (GAG), the only one that exists free, not bound to proteins.
Sodium hyaluronate is made of disaccharide units consisting of sodium glucuronate and N-acetylglucosamine. It is present in all tissues, is highly biocompatible and forms the main component of synovial fluid, where it is found in high concentration.
Mechanism of action of sodium hyaluronate:
Viscosupplementation → enables lubrication and shock absorption
Visco-induction → Establishing joint homeostasis by inducing endogenous production of hyaluronic acid lasts a long time after the injection itself
The most important effects of sodium hyaluronate:
- Lubricant effect
- Anti-inflammatory effect
- Analgesic effect
- Chondroprotective effect
- Endogenous synthesis of hyaluronic acid
Clinical studies have proven that the intra-articular application of sodium hyaluronate provides a reduction in the simultaneous use of analgesics by up to 50%, and postpones the need for surgical implantation of a prosthesis.
Chondroitin sulfate
Chondroitin sulfate belongs to glycosaminoglycans (GAG), of a high molecular weight (10,000 - 50,000 Da), is made of disaccharide units consisting of sodium glucuronate and N-acetylgalactosamine.
Chondroitin sulfate is found in animal and fish cartilage in food, while in humans this substance is mostly synthesized by chondrocytes of long bones, incorporated into complex proteoglycans and excreted into the extracellular matrix of bones.
Chondroitin sulfate is necessary for maintaining the structural integrity of tissues, primarily skin and cartilage.
Deficiency of chondroitin sulfate in cartilage is associated with the onset of osteoarthritis.
The most important effects of chondroitin sulfate:
- Optimizes the rheological behavior of hyaluronic acid (due to specific interactions)
- It inhibits metalloproteinases and aggrecanases, enzymes that break down cartilage
- Anti-inflammatory effect (reduces the secretion of pro-inflammatory factors)
- Pain reduction
- Improves joint functionality
Clinical studies have confirmed the effectiveness of the combined use of sodium hyaluronate and chondroitin sulfate in the treatment of osteoarthritis.
N Acetyl Glucosamine (NAG)
N acetyl - glucosamine (NAG) is a monosaccharide derivative of glucose. It is a naturally occurring compound found in the exoskeletons of crustaceans, in the cell walls of fungi and bacteria, and in human cartilage.
NAG is the basic building block of complex glycosaminoglycans of cartilage and connective tissue.
NAG is useful in preventing joint damage; it has a stimulating effect on the synthesis of hyaluronic acid in chondrocytes.
Instructions for use (pdf 1.02 MB)
Write to us
Mosorska 9, 11000 Belgrade, Serbia
Vemax011 Pharma doo representative office Northern Macedonia - Dane Krapcev 13, Skopje
Vemax011 Pharma doo representative office Montenegro - Topliški put 1, Budva
Vemax011 Pharma doo representative office Bosnia and Hercegovina - Vlakovo 252, Sarajevo
+381 (0)63 103 00 08
office@vemaxpharma.rs
Vemax Pharma - your trusted regional partner on the road to health!
